 标准还原电势StandardReductionPotential范本模板文档格式.docx
标准还原电势StandardReductionPotential范本模板文档格式.docx
- 文档编号:17014947
- 上传时间:2022-11-27
- 格式:DOCX
- 页数:28
- 大小:25.55KB
标准还原电势StandardReductionPotential范本模板文档格式.docx
《标准还原电势StandardReductionPotential范本模板文档格式.docx》由会员分享,可在线阅读,更多相关《标准还原电势StandardReductionPotential范本模板文档格式.docx(28页珍藏版)》请在冰豆网上搜索。
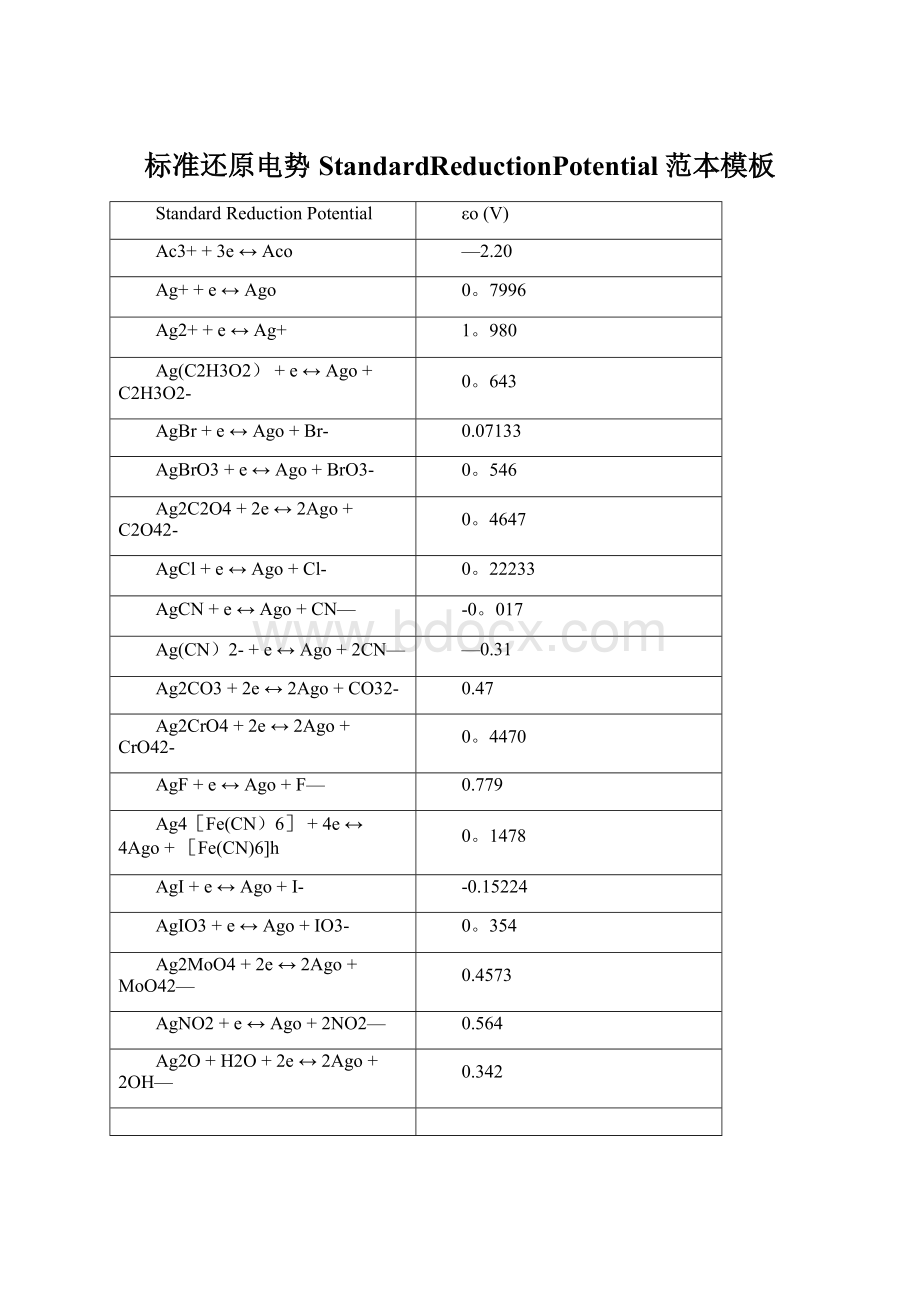
08951
Ag2S+2H++2e↔2Ago+H2S
0366
Ag2SeO3+2e↔2Ago+SeO42-
3629
Ag2WO4+2e↔2Ago+WO42-
4660
Ag2SO4+2e↔2Ago+SO42-
0.654
Ag(S2O3)23—+e↔Ago+2S2O32-
01
Al3++3e↔Alo
-1。
662
Al(OH)3+3e↔Alo+3OH-
-2。
31
Al(OH)4—+3e↔Alo+4OH—
—2.328
H2AlO3—+H2O+3e↔Alo+4OH-
—2.33
AlF63—+3e↔Alo+6F-
069
Am4++e↔Am3+
2.60
Am2++2e↔Amo
—1。
Am3++3e↔Amo
—2.048
Am3++e↔Am2+
—2.3
Aso+3H++3e↔AsH3
—0。
608
As2O3+6H++6e↔2Aso+3H2O
234
HAsO2+3H++3e↔Aso+2H2O
248
AsO2-+2H2O+3e↔Aso+4OH—
68
H3AsO4+2H++2e↔HAsO2+2H2O
0.560
AsO43-+2H2O+2e↔AsO2-+4OH—
—0.71
Aso+3H2O+3e↔AsH3+3OH—
21
At2+2e↔2At—
0.3
Au++e↔Auo
1.692
Au3++2e↔Au+
1.401
Au3++3e↔Auo
1.498
Au2++e↔Au+
1.8
AuOH2++H++2e↔Au++H2O
32
AuBr2—+e↔Auo+2Br—
0.959
AuBr4—+3e↔Auo+4Br-
854
AuCl4-+3e↔Auo+4Cl-
002
Au(OH)3+3H++3e↔Auo+3H2O
1.32
H2BO3—+5H2O+8e→BH4-+8OH-
24
H2BO3—+H2O+3e→Bo+4OH-
—1.79
H3BO3+3H++3e→Bo+3H2O
8698
B(OH)3+7H++8e→BH4—+3H2O
—0.481
Ba2++2e→Bao
—2.912
Ba2++2e→Ba(Hg)
570
Ba(OH)2+2e→Bao+2OH—
99
Be2++2e→Beo
-1.847
Be2O32-+3H2O+4e→2Beo+6OH—
—2.63
p-benzoquinone+2H++2e→hydroquinone
6992
Bi++e→Bio
0.5
Be3++3e→Bio
0.308
Bi3++2e→Bi+
2
Bio+3H++3e→BiH3
BiCl4—+3e→Bio+4Cl—
16
Bi2O3+3H2O+6e→2Bio+6OH—
46
Bi2O4+4H++2e→2BiO++2H2O
1.593
BiO++2H++3e→Bio+H2O
320
BiOCl+2H++3e→Bio+Cl-+H2O
1583
Bk4++e→Bk3+
1.67
Bk2++2e→Bko
67
Bk3++e→Bk2+
Br2(aq)+2e→2Br—
1.0873
Br2(l)+2e→2Br—
066
HBrO+H++2e→Br-+H2O
1.331
HBrO+H++e→1/2Br2(aq)+H2O
1.574
HBrO+H++e→1/2Br2(l)+H2O
1.596
BrO-+H2O+2e→Br-+2OH-
0.761
BrO3—+6H++5e→1/2Br2+3H2O
1.482
BrO3—+6H++6e↔Br—+3H2O
1.423
BrO3—+3H2O+6e↔Br-+6OH—
61
BrO—+H2O+e↔1/2Br2+4OH—
0.455
(CN)2+2H++2e↔2HCN
373
2HCNO+2H++2e↔(CN)2+2H2O
0.330
(CNS)2+2e↔2CNS—
77
CO2+2H++2e↔HCOOH
199
HCHO+2H2O+2e↔CH3OH+2OH-
-0.59
2CO2+2H++2e↔H2C2O4
—0.49
C2N2+2H++2e↔2HCN
0.37
Ca++e↔Cao
—3。
80
Ca2++2e↔Cao
—2。
868
Ca(OH)2+2e↔Cao+2OH-
02
Cd2++2e↔Cdo
4030
Cd2++2e↔Cd(Hg)
—0.3521
Cd(OH)2+2e↔Cd(Hg)+2OH—
-。
809
CdSO4+2e↔Cdo+SO42-
—0.246
Cd(OH)42-+2e↔Cdo+4OH—
658
CdO+H2O+2e↔Cdo+2OH—
—0.783
CdS+2e↔Cdo+S2—
-1.21
Ce3++3e↔Ceo
-2.336
Ce3++3e↔Ce(Hg)
4373
Ce4++e↔Ce3+
72
CeOH3++H++e↔Ce3++H2O
715
Cf4++e↔Cf3+
3.3
Cf3++e↔Cf2+
6
Cf3++3e↔Cfo
—1.94
Cf2++2e↔Cfo
—2.12
Cl2(g)+2e↔2Cl—
1.35827
HClO+H++e↔1/2Cl2+H2O
1.611
HClO+H++2e↔Cl-+H2O
482
ClO-+H2O+2e↔Cl-+2OH-
0.81
ClO2+H++e↔HClO2
1.277
HClO2+2H++2e↔HClO+H2O
645
HClO2+3H++3e↔1/2Cl2+2H2O
628
HClO2+3H++4e↔Cl-+2H2O
1.570
ClO2—+H2O+2e↔ClO-+2OH-
0.66
ClO2-+2H2O+4e↔Cl—+4OH—
76
ClO2(aq)+e↔ClO2—
0.954
ClO3—+2H++e↔ClO2+H2O
152
ClO3-+3H++2e↔HClO2+H2O
1.214
ClO3—+6H++5e↔1/2Cl2+3H2O
1.47
ClO3-+6H++6e↔Cl-+3H2O
1.451
ClO3—+H2O+2e↔ClO2—+2OH—
0.33
ClO3—+3H2O+6e↔Cl-+6OH-
62
ClO4—+2H++2e↔ClO3—+H2O
1.189
ClO4—+8H++7e↔1/2Cl2+4H2O
1.39
ClO4—+8H++8e↔Cl-+4H2O
1.389
ClO4-+H2O+2e↔ClO3—+2OH—
0.36
Cm4++e↔Cm3+
3.0
Cm3++3e↔Cmo
04
Co2++2e↔Coo
28
Co3++e↔Co2+
92
[Co(NH3)6]3++e↔[Co(NH3)6]2+
108
Co(OH)2+2e↔Co+2OH-
—0.73
Co(OH)3+e↔Co(OH)2+OH—
0.17
Cr2++2e↔Cro
913
Cr3++e↔Cr2+
-0.407
Cr3++3e↔Cro
-0.744
Cr2O72-+14H++6e↔2Cr3++7H2O
1.232
CrO2—+2H2O+3e↔Cro+4OH-
Cr5++e↔Cr4+
1.34
HCrO4—+7H++3e↔Cr3++4H2O
350
CrO42—+4H2O+3e↔Cr(OH)3+5OH—
-0.13
Cs++e↔Cso
026
Cu++e↔Cuo
0.521
Cu2++e↔Cu+
153
Cu2++2e↔Cuo
0.3419
Cu2++2e↔Cu(Hg)
345
Cu3++e↔Cu2+
2.4
Cu2O3+6H++2e↔2Cu2++3H2O
2.0
Cu2++2CN—+e↔[Cu(CN)2]-
1.103
CuI2-+e↔Cuo+2I-
00
CuI+e↔Cuo+I—
—0.185
Cu2++I—+e↔CuI
0.86
Cu2O+H2O+2e↔2Cuo+2OH—
360
Cu(OH)2+2e↔Cuo+2OH-
-0.222
2Cu(OH)2+2e↔Cu2O+2OH-+H2O
080
-2.923
2D++2e↔D2
013
Dy2++2e↔Dyo
Dy3++3e↔Dyo
-2.295
Dy3++e↔Dy2+
Er2++2e↔Ero
—2.0
Er3++3e↔Ero
-2.331
Er3++e↔Er2+
-3。
Es3++e↔Es2+
3
Es3++3e↔Eso
—1.91
Es2++2e↔Eso
23
Eu2++2e↔Euo
812
Eu3++3e↔Euo
—1.991
Eu3++e↔Eu2+
-0.36
F2+2H++2e↔2HF
3.053
F2+2e↔2F-
2.866
F2O+2H++4e↔H2O+2F—
2。
153
Fe2++2e↔Feo
447
Fe3++3e↔Feo
—0.037
Fe3++e↔Fe2+
771
2HFeO4—+8H++6e↔Fe2O3+5H2O
09
HFeO4—+4H++3e↔FeOOH+2H2O
08
HFeO4—+7H++3e↔Fe3++4H2O
07
Fe2O3+4H++2e↔2FeOH++H2O
0.16
[Fe(CN)6]3-+e↔[Fe(CN)6]4-
0.358
FeO42—+8H++3e↔Fe3++4H2O
2.20
Fe(OH)3+e↔Fe(OH)2+(OH)-
56
FeS+2e↔Feo+S2-
-1.01
Fe(OH)2+2e↔Feo+2OH-
—0.877
Fm3++e↔Fm2+
-1.1
Fm3++3e↔Fmo
—1.89
Fm2++2e↔Fmo
—2.30
Fr++e↔Fro
—2.9
Ga3++3e↔Gao
-0.549
Ga++e↔Gao
-0.2
GaOH2++H++3e↔Gao+H2O
—0.498
H2GaO3—+H2O+3e↔Gao+4OH-
—1.219
Gd3++3e↔Gdo
279
Ge2++2e↔Geo
Ge4++4e↔Geo
0.124
Ge4++2e↔Ge2+
0.00
GeO2+2H++2e↔GeO+H2O
-0.118
H2GeO3+4H++4e↔Geo+3H2O
182
2H++2e↔H2
0.0000000
H2+2e↔2H—
-2.23
HO2+H++e↔H2O2
1.495
2H2O+2e↔H2+2OH-
8277
H2O2+2H++2e↔2H2O
1.776
H2O2+2e↔2OH-
0.88
HO2-+H2O+2e↔3OH—
88
Hf4++4e↔Hfo
-1.55
HfO2++2H++4e↔Hfo+H2O
724
HfO2+4H++4e↔Hfo+2H2O
—1.505
HFO(OH)2+H2O+4e↔Hfo+4OH—
-2.50
Hg2++2e↔Hgo
0.851
2Hg2++2e↔Hg22+
920
Hg22++2e↔2Hgo
0.7973
Hg2(C2H3O2)2+2e↔2Hgo+2(C2H3O2)-
51163
Hg2Br2+2e↔2Hg+2Br—
13923
Hg2Cl2+2e↔2Hgo+2Cl—
26808
Hg2HPO4+2e↔2Hgo+HPO42-
6359
Hg2I2+2e↔2Hgo+2I—
0405
Hg2O+H2O+2e↔2Hgo+2OH-
0.123
HgO+H2O+2e↔Hgo+2OH-
0977
Hg(OH)2+2H++2e↔Hgo+2H2O
1.034
Hg2SO4+2e↔2Hgo+SO42-
0.6125
2HgCl2+2e↔Hg2Cl2+2Cl-
63
HO2++2e↔Ho
1
Ho3++3e↔Ho
I2+2e↔2I—
0.5355
I3-+2e↔3I-
536
H3IO62-+2e↔IO3—+3OH-
0.7
H5IO6+H++2e↔IO3—+3H2O
601
2HIO+2H++2e↔I2+2H2O
1.439
HIO+H++2e↔I—+H2O
0.987
IO-+H2O+2e↔I-+2OH-
0.485
2IO3-+12H++10e↔I2+6H2O
195
IO3—+6H++6e↔I-+3H2O
1.085
IO3—+2H2O+4e↔IO—+4OH—
0.15
IO3-+3H2O+6e↔IO—+6OH—
26
IO4-+2H++2e↔IO3—+H2O
60
2IO-+2H2O+2e↔I2+4OH—
42
In++e↔Ino
-0.14
In2++e↔In+
40
In3++e↔In2+
49
In3++2e↔In+
-0.443
In3++3e↔Ino
3382
In(OH)3+3e↔Ino+3OH—
—0.99
In(OH)4-+3e↔Ino+4OH-
—1.007
In2O3+3H2O+6e↔2Ino+6OH-
034
Ir3++3e↔Iro
156
[IrCl6]2-+e↔[IrCl6]3-
0.8665
[IrCl6]3-+3e↔Iro+6Cl—
0.77
Ir2O3+3H2O+6e↔2Iro+6OH-
0.098
K++e↔Ko
931
La3++3e↔Lao
379
La(OH)3+3e↔Lao+3OH—
90
Li++e↔Lio
—3.0401
Lr3++3e↔Lro
-1.96
Lu3++3e↔Luo
—2.28
Md3++e↔Md2+
Md3++3e↔Mdo
65
Md2++2e↔Mdo
Mg++e↔Mgo
70
Mg2++2e↔Mgo
372
Mg(OH)2+2e↔Mgo+2OH-
690
Mn2++2e↔Mno
—1.185
Mn3++3e↔Mn2+
1.5415
MnO2+4H++2e↔Mn2++2H2O
224
MnO4—+e↔MnO42—
558
MnO4—+4H++3e↔MnO2+2H2O
1.679
MnO4—+8H++5e↔Mn2++4H2O
507
MnO4-+2H2O+3e↔MnO2+4OH—
595
MnO42—+2H2O+2e↔MnO2+4OH-
Mn(OH)2+2e↔Mno+2OH—
—1.56
Mn(OH)3+e↔Mn(OH)2+OH—
15
Mn2O3+6H++e↔2Mn2++3H2O
MnO2+2H2O+2e↔Mn(OH)2+2OH—
05
Mo3++3e↔Moo
-0.200
MoO2+4H++4e↔Moo+4H2O
H3Mo7O243—+45H++42e↔7Moo+24H2O
0.082
MoO3+6H++6e↔Moo+3H2O
075
N2+2H2O+6H++6e↔2NH4OH
092
3N2+2H++2e↔2HN3
-3.09
N5++3H++2e↔2NH4+
1.275
N2O+2H++2e↔N2+H2O
766
H2N2O2+2H++2e↔N2+2H2O
2.65
N2O4+2e↔2NO2-
0.867
N2O4+2H++2e↔2NHO2
065
N2O4+4H++4e↔2NO+2H2O
1.035
2NH3OH++H++2e↔N2H5++2H2O
1.42
2NO+2H++2e↔N2O+H2O
- 配套讲稿:
如PPT文件的首页显示word图标,表示该PPT已包含配套word讲稿。双击word图标可打开word文档。
- 特殊限制:
部分文档作品中含有的国旗、国徽等图片,仅作为作品整体效果示例展示,禁止商用。设计者仅对作品中独创性部分享有著作权。
- 关 键 词:
- 标准 还原 电势 StandardReductionPotential 范本 模板
 冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。
冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。


 如何打造酒店企业文化2刘田江doc.docx
如何打造酒店企业文化2刘田江doc.docx
 地下车库顶种植屋面标准做法分析100117.ppt
地下车库顶种植屋面标准做法分析100117.ppt
