 电化学与热力学Electrochemistry and Thermodynamics.docx
电化学与热力学Electrochemistry and Thermodynamics.docx
- 文档编号:10007129
- 上传时间:2023-02-07
- 格式:DOCX
- 页数:11
- 大小:74.86KB
电化学与热力学Electrochemistry and Thermodynamics.docx
《电化学与热力学Electrochemistry and Thermodynamics.docx》由会员分享,可在线阅读,更多相关《电化学与热力学Electrochemistry and Thermodynamics.docx(11页珍藏版)》请在冰豆网上搜索。
电化学与热力学ElectrochemistryandThermodynamics
E10
ElectrochemistryandThermodynamics
E10ElectrochemistryandThermodynamics
INTRODUCTION
Inthisexperimentyouwillstudytheelectrochemistryofvoltaic(galvanic)cellsandintheprocessdeterminethevaluesofthethermodynamicfunctions∆G,∆H,and∆S.Youwillconstructelectrochemicalcellsbycombiningdifferentmetallicsystemsandtheirsolutions.Measuringthepotentialofthepreparedcellsatvarioustemperatureswillrenderthevaluesofthethermodynamicfunctions∆G,∆H,and∆Scorrespondingtotheelectrochemicalsystemstudied.
THEORETICALBACKGROUND
Electrochemistry:
Electrochemistryistheareaofchemistrythatdealswiththerelationbetweenchemicalchangesandelectricalenergy.Chemicalreactionscanbeusedtoproduceelectricalenergyinvoltaic(galvanic)cells.Electricalenergy,ontheotherhandcanbeusedtobringaboutchemicalchangesinwhataretermedelectrolyticcells.Inthisexperimentyouwillinvestigatesomeofthepropertiesofvoltaiccells.
Oxidation-reductionreactionsarethosethatinvolvethetransferofelectronsfromonesubstancetoanother.Aredoxreactionisthesimultaneousoccurrenceofthetwocomponentsorhalfreactions.Oxidationoccurswhenachemicalspecieslosesorgivesupelectronstoanotherchemicalspecies.Reductionoccurswhenachemicalspeciesreceivesorgainelectrons.Theoxidationprocessprovidestheelectronsnecessaryforreductiontooccur.Therefore,theoxidizedspeciesisthereducingagent.,andthereducedspeciesistheoxidizingagent.
Forexample,ifapieceofzincmetalisimmersedintoasolutioncontainingcopper(II)ions,zincwillbeoxidizedbythecopper(II)ions.Zincloseselectronsanditisoxidizedwhilecopper(II)ionsgainelectronsandarereduced.Wecanconvenientlyexpressthesetwoprocessesbythefollowingtwohalf-reactions,whichaddtogivetheoverallredoxreaction.
Thefunctionofavoltaiccellisbaseduponreactionssimilartotheoneillustratedinequation
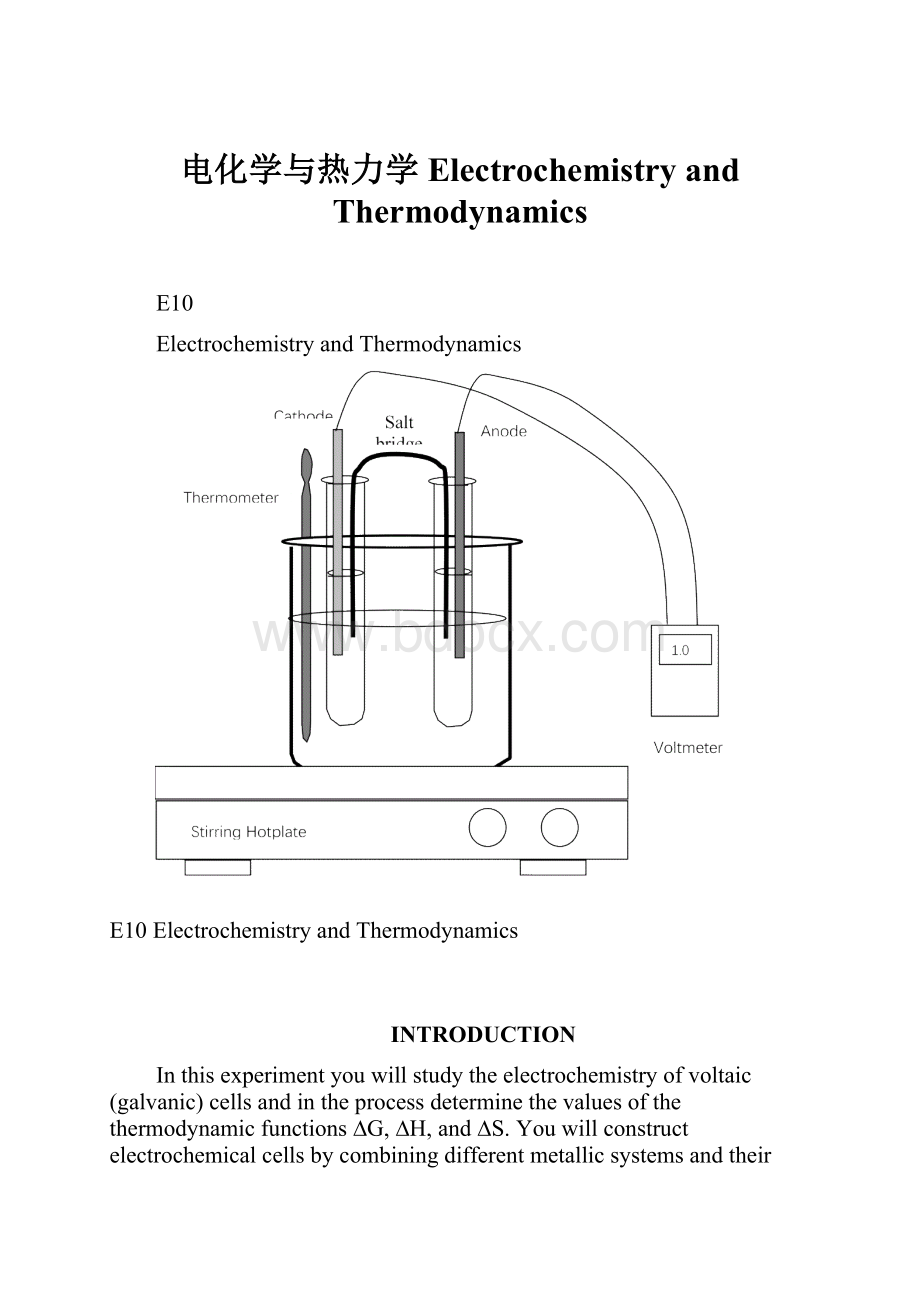
(1).ThesystemZn/CuiscalledtheDaniellcellinhonorofJohnFrederickDaniell(1790-1845)whodevelopedthecellforthefirsttime.Asaltbridgemustbeusedtoavoidpolarizationoftheelectrodesbyfacilitatingthecirculationofionsfrombothcellcompartments.(seefigure1).
Figure1ElectrochemicalCell
Thecellvoltage,ortheelectromotiveforce(abbreviatedemf),isindicatedonthevoltmeterinvolts.Thecellemfisalsocalledthecellpotential.Themagnitudeoftheemfisaquantitativemeasureofthedrivingforceorthermodynamictendencyforthereactiontooccur.Ingeneral,theemfofavoltaiccelldependsuponthesubstancesthatmakeupthecellaswellasontheirconcentrations.Hence,itiscommonpracticetocomparestandardcellpotentials,symbolizedbyEocell.Thesepotentialscorrespondtocellvoltagesunderstandardstateconditions-gasesat1atmpressure,solutionsat1Mconcentration,andtemperatureat25oC.
Justastheoverallcellreactionmayberegardedasthesumoftwohalf-reactionstheoverallcellemfcanbethoughtofasthesumoftwohalf-cellpotentials,thatis,thesumofthevoltageoftheoxidationhalf-reaction(Eo(ox))andthevoltageofthereductionhalf-reaction(Eo(red))
(2)
Becauseitisimpossibletomeasuredirectlythepotentialofanisolatedhalf-cell,thestandardhydrogenhalf-reactionhasbeenselectedasareferenceandhasbeenassignedastandardreductionpotentialofexactly0.000V;
(3)
Let’slookagainattheDaniellcelldescribedabove.Thestandardhalf-cellpotentialsare:
Therefore,
(4)
Thecompletenotationschemeforagalvaniccelliswrittenwiththeanodeontheleftandadoubleverticallinedenotingthepresenceofaporousplateorsaltbridge.FortheDaniellcell:
Zn|Zn2+||Cu2+|Cu.
Eachelectrodeisconnectedtothevoltmeterbyalligatorclipsandmetalwiring.Thevoltmetermeasuresthevoltagegeneratedbytheredoxreaction.Thevoltagereadingwillbepositivewhentheelectrodesareconnectedproperlyforspontaneousreaction.Aspontaneousredoxreactionoccurswhenthespecieswithhigherreductionpotentialisconnectedasthecathode.Otherwise,thevoltagereadingwillbenegative.Themeterreadingwillbepositivewhenthecathodeisconnectedtothe(+)outletandtheanodeisconnectedtothe(-)outlet.Physically,anegativevoltagereadingmeansthatyouhaveconnectedthewrongelectrodeascathode.Thisisequivalenttoreversingequation
(1).Whenequation
(1)isreversed,themeasuredcellpotentialdifferencebecomes
Eocell=-1.10V.Theabsolutevalueof∆Eocellisthesameinbothcases,butthesignisdifferent.Thesignof∆Eocellispositiveforaspontaneousreactionandnegativefornon-spontaneousreaction.
TheNernstEquation:
Formeasurementstakenunderstandardconditions(1atm,1Msolutions),∆Eocellmeasurestheelectricpotentialdifferencebetweenthehalf-cells.Formeasurementstakenundernon-standardconditions(theusuallaboratorysituation),theNernstequationisusedtocalculate∆Ecell.TheNernstequationgivesustherelationshipbetweentheoverallcellpotentialdifferenceforaredoxreaction∆Ecellandtheconcentrationsofthemetal-ionsolutions.TheNernstequationis;
(5)
whereFisFaradayconstant.Rtheuniversalgasconstant,nthenumberofelectronstransferred,andQisthereactionquotient.Whennatural(ln)isconvertedtobaseten(log)and(RT/nF)areevaluatedusingR=8.315JK-1mol-1,T=295.15K,andF=96,485Cmol-1theequationbecomes;
(6)
Inthisform,galvaniccellsareusedtodeterminetheconcentrationofthemetalionspresentunderconditionsotherthanstandardconditions.IfweapplytheNernstequationtoequation
(1)wewillget
(7)
Thermodynamics:
Morethanacenturyago,scientistsobservedtheeffectoftemperatureonnaturalprocessesandformulatedthelawsofthermodynamics.Thermodynamicsisthestudyoftheflowofenergy,especiallyintheformofheat,anditsconversionfromoneformtoanother(forexample,fromchemicalenergytoheatenergy).Thelawsofthermodynamicsareasetofmathematicalfunctions,whichgovernallformsofenergyandtheirinter-conversion.
Thezerothlawofthermodynamicsisconcernedwithtemperatureequilibrium;itstatesthatheatalwaysflowsfromahottoacoldbody.
Thefirstlawofthermodynamicsdescribestheconservationofenergy.Itstatesthatenergycannotbecreatedordestroyed;itcanonlybeconvertedfromoneformtoanother.
Thesecondlawofthermodynamicsisconcernedwiththereversibilityanddirectionofnaturalevents;itstatesthattheentropyoftheuniversetendstoincrease.Therefore,allspontaneouseventsareaccompaniedbyanincreaseintheentropyoftheuniverse.Entropy(S)isameasureofdisorder.Thehighertheentropyofasystem,thelowertheorderofthatsystem.Forexample,theentropyofheliumsealedinsideaballoonislowerthanitsentropywhenisreleasedtoopenspace.Whenintheballoon,theindividualheliumatomsareconfinedtothespacewithintheballoon.Whenthegasisreleasedtheatomsrushoutintoalessorderedstate.
TheThirdlawdefinesthezeroofentropyforasystem.Itstatesthatpuresubstancesatabsolutezero(0Kelvin),thetemperatureatwhichallatomicmotionceases,haveentropyofzero.
Whentheselawsareappliedtochemicalreactions,theyrelatetheequilibriumcondition(whetherthereactionisproceedingintheforwardorreversedirection)andthetemperatureofthereactiontothethermodynamicfunctions,namelychangeinenthalpy(∆H),changeinentropy(∆S),andchangeinfreeenergy(∆G).Bystudyingtheeffectoftemperatureontheequilibriumconditionforachemicalreaction,wecancalculatethesethermodynamicfunctions.
Byexamining∆H,∆Sand∆Ginachemicalsystem,awealthofinformationmaybeextracted.Inthermodynamicsasystemistheparticularpartoftheuniversebeingstudied.
Enthalpydescribeschangesofheatinasystem.Ifthesystemabsorbsheatduringagivenprocess,thisprocessissaidtobeendothermic,andthechangeinenthalpy(∆H)isgreaterthanzero(H>0).Onthecontrary,ifthesystemreleasesheat,theprocessiscalledexothermic,and(∆H<0).
Asmentionedabove,entropyisthemeasureofdisorderorrandomnessinasystem.Thegreatertheorderinasystem,thelowertheentropy.Thelowertheorderinasystem,thehighertheentropy.
TheGibbsFreeEnergy(G)isthefunctionderivedfromHandS.Thechangeinfreeenergy(∆G)isacompositefunctionthatgivesameasureofthespontaneityofareaction.If∆G<0,areactionwilloccurspontaneously.If∆G>0,thereactionisnotspontaneous.If∆G=0thesystemisatequilibrium.Themathematicalformof∆Gisthefollowing:
(8)
WhereTistheabsolutetemperature(inKelvin).Theeffectofthesignof∆Hand∆Sandtheeffectoftemperatureonspontaneityofareactioncanbesummarizedasfollows:
∆Hsystem
∆Ssystem
Process
-
+
Spontaneousatalltemperatures
+
-
Non-spontaneousatalltemperatures
+
+
Spontaneousathightemperatures
-
-
Spontaneousatlowtemperatures
ThermodynamicsandEquilibrium:
ForageneralreactionorprocessofAandBgivingproductsCandD:
thereactionquotient,Q,is:
(9)
The∆GofaprocessatanyconcentrationofA,B,C,andD,isgivenby:
(10)
Where∆Goisthefreeenergyofthereactionunderthedefinedstandardconditionsof1atmand1Mconcentrationsofreactants.∆Gistheobservedchangeinfreeenergyatconditionsotherthanstandard.AtequilibriumQ=Keq.Thereactionfavorsneitherproductsnorreactantsatequilibrium,a
- 配套讲稿:
如PPT文件的首页显示word图标,表示该PPT已包含配套word讲稿。双击word图标可打开word文档。
- 特殊限制:
部分文档作品中含有的国旗、国徽等图片,仅作为作品整体效果示例展示,禁止商用。设计者仅对作品中独创性部分享有著作权。
- 关 键 词:
- 电化学与热力学Electrochemistry and Thermodynamics 电化学 热力学 Electrochemistry
 冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。
冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。


 1212中级汽车维修工考试试题三.docx
1212中级汽车维修工考试试题三.docx
